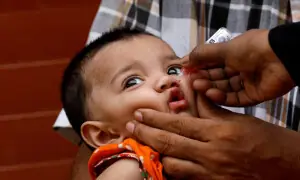
Breakthrough malaria vaccine offers to reinvigorate the fight against the disease
4 min readThe World Health Organization has announced a historic move: it has recommended the widespread use of the first ever malaria vaccine. The recommendation is based on the results of an ongoing pilot programme in Malawi, Ghana and Kenya. Malaria is a huge global health challenge, around 409,000 people died of malaria in 2019 alone. The WHO African region carries significant proportion of the malaria burden – 94% of all malaria cases and deaths occurred in the region. Children younger than five are the most vulnerable. Ina Skosana asked Eunice Anyango Owino to explain the development, and its signficance.
The most significant reason is that the malaria parasite is very complex. It has different stages; some in the mosquito and some in the human. Thus, scientists had to pursue a diversity of approaches.
For example, in the human there are two stages. These are the:
Pre-erythrocytic stages (sporozoite, or spore-like, stage). This is the period when the parasite’s sporozoite from a mosquito bite enters the blood stream and heads for the liver to mature and multiply after which they are then released.
The blood stage (Merozoite stage). This is when the parasite’s merozoites are released from the liver, and multiply in the red blood cells.
So an effective vaccine against the first stage (pre-erythroctic stage) would be one that elicits an immune response that would either prevent infection of the liver cells or lead to destruction of the infected liver cells.
An effective vaccine for the second stage (blood stage) would be the one that does one of three things: elicits immune responses that prevent infection of red blood cells; decreases the number of parasites in the blood; reduces the severity of the disease by allowing the body to develop a natural immunity with little risk of getting ill.
Another option was to develop transmission blocking vaccines. An effective transmission blocking vaccine would induce antibodies that would block maturation of malaria parasites in mosquitoes that feed on vaccinated individuals.
Another factor that contributed to the delay is that scientists working on malaria vaccines in the early stages lacked an understanding of the specific immune responses associated with protection against the parasite.
Also, malaria parasites – such as Plasmodium falciparum – display a variety of antigens on their surfaces that help them escape the immune system and also render vaccines based on specific antigens less effective.
The vaccine RTS,S, trade name Mosquirx, is given in four doses to children from 5 months old to 17 months; the first 3 doses are given monthly with the first at 5 months and the third at 9 months. The fourth, which is a booster dose, is given at between 15 and 18 months.
The efficacy is about 40% against malaria cases and 30% against severe malaria.
No two vaccines or diseases are comparable. The WHO has set an efficacy level of 50% and above for most vaccines and most highly efficacious vaccines offer 75 % and above level of protection. For example, the COVID-19 vaccines based on messenger (m)RNA technology by Pfizer and Moderna offer well above 90% protection level.
The RTS,S vaccine targets the parasites before they infect liver cells; it targets the circumsprorozoite protein on P.falciparum sporozoites surface and is thus considered a pre-erythrocytic vaccine.
First, the WHO and the manufacturers of the vaccine GlaxoSmithKline will be rallying countries, particularly those with high malaria burdens, to adopt the vaccine as part of their National Malaria Control Strategies.
They will also be asking these countries to set aside funds.
They will also be involved in fundraising from the global health community or work with partners, for a broader roll out of the vaccine.
There should be an equitable and long term access to the vaccine. The vaccine should also be cost effective.
The hope is that the WHO announcement will re-energise the race to find even more efficient vaccines against malaria. Current reports by the Jenner Institute of Oxford University suggest that a malaria vaccine that might meet the WHO goal of 75% is under trial in Burkina Faso.
The vaccine is an additional tool to the malaria control tool kit.
The vaccine does not provide complete protection. And will be introduced as part of a tool kit geared to reducing malaria infections and reducing fatalities. Other measures include bed nets and indoor residual spraying.
Nevertheless the vaccine has a great potential to reduce death and illness in high burden areas in sub-Saharan Africa especially if used in combination with pre-existing malaria prevention methods. For example a study by the London School of Tropical Medicine reported a 70% reduction in hospitalisations and death in children given the mosquirx vaccine plus antimalarial drugs.
Malaria control had been stagnating in some African countries, with countries like Sudan and Eritrea seeing a significant resurgence in the recent past.
The vaccine will reinvigorate the fight against malaria. And offers the promise of bringing it back on track.
For the latest news, follow us on Twitter @Aaj_Urdu. We are also on Facebook, Instagram and YouTube.
























Comments are closed on this story.